How tissue homeostasis is maintained during physiological conditions or upon tissue damage remains the top important yet unresolved issue. Accumulating evidence over the past two decades has established the Hippo signaling pathway as central regulator of organ size control, tissue homeostasis and regeneration. Excessive activation or absence of the Hippo signaling leads to abnormal tissue growth, disrupted tissue regeneration and repair that ultimately result in tissue damage and tumorigenesis. The core members of this pathway are composed of the upstream kinase complex MST/SAV/LATS/MOB and the downstream transcriptional complex YAP/TAZ-TEADs. Despite the well-defined regulatory mechanism of Hippo signaling activity, the molecular basis that tissue/organ initiates specific target gene program to orchestrate tissue homeostasis is unclarified. Particularly, how the Hippo signaling pathway regulates cell apoptosis is in need of further study.
The study entitled as “A regulation loop between YAP and NR4A1 balances cell proliferation and apoptosis” was published in Cell Reports on October 20, 2020.
By integrating genome-wide multi-omics approach, researchers led by Prof. ZHANG Lei from the Center for Excellence in Molecular Cell Science, Shanghai Institute of Biochemistry and Cell Biology of the Chinese Academy of Sciences (CAS) has identified NR4A1 as a key mediator of cell apoptosis significantly repressed by YAP via recruiting the epigenetic complex NuRD. In addition, YAP constrained mitochondrial localization of NR4A1 through AKT to abrogate its proapoptotic function. As a feedback loop, NR4A1 facilitates YAP ubiquitination and degradation to augment the Hippo signaling. This reciprocal relationship of YAP/NR4A1 is of physiological significance during liver regeneration and tumorigenesis. The repression of NR4A1 upon partial hepatectomy is critical to establish YAP signaling robustness for efficient hepatocyte proliferation and liver regeneration. Activation of NR4A1 suppresses YAP activity to avoid tissue overgrowth that lead to tumorigenesis, where YAP is commonly overexpressed. Collectively, this study identifies NR4A1 as a directly repressed target of YAP/TAZ-TEADs and show that the reciprocal feedback regulation between YAP and NR4A1 enables proper organ size during liver regeneration and constrains tumorigenesis to maintain tissue homeostasis.
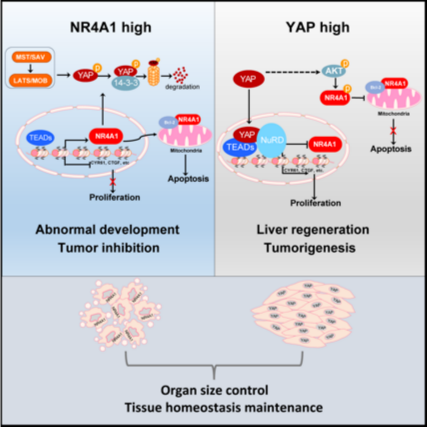
Proposed working model of the regulation loop between YAP and NR4A1:Under the circumstance of environmental stresses, such as liver damage that initiating liver regeneration or tumor initiation, inactivation of Hippo signaling, on the one hand, triggers YAP activation and nuclear translocation to recruit NuRD complex, leading to transcriptional repression of NR4A1 and activation of proliferation-related target genes. On the other hand, activated YAP signaling drives AKT-dependent phosphorylation of NR4A1 to disrupt its mitochondrial localization and pro-apoptotic activity. These events are critical to establish YAP signaling robustness for efficient hepatocyte proliferation and liver regeneration. When liver normally reaches its approximate size, the Hippo signaling is reactivated to restore the expression and mitochondrial targeting of NR4A1 to help constrain YAP activity through enhancing its binding to 14-3-3 and degradation, which consequently results in proper tissue homeostasis to avoid tissue overgrowth that lead to tumorigenesis, where YAP is commonly overexpressed.
Contact: rayzhang@sibcb.ac.cn
Reference: https://www.cell.com/cell-reports/fulltext/S2211-1247(20)31273-0
 Appendix:
Appendix: