A cation-π interaction regulates ligand binding affinity and signaling of integrin α4β7
Source:
Time: 2010-11-26
On November 22th, the Proceedings of the National Academy of Sciences online published the latest findings of Prof. Jianfeng Chen’s Lab from SIBCB. The research team discovered that a novel cation-π interaction playing vital roles in the regulation of integrin affinity, signaling and biological functions.
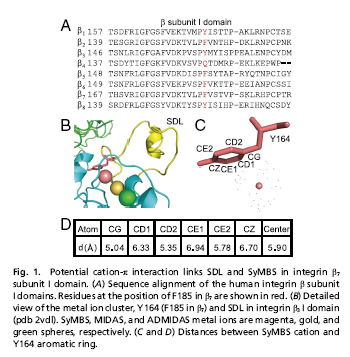
Integrin α4β7 mediates rolling and firm adhesion of leucocytes, two of the critical steps in leukocyte migration and tissue specific homing. Affinity of α4β7 for ligand is dynamically regulated by three interlinked metal ion-binding sites in β7-subunit I domain. In this study, the authors found that Phe185 (F185), a highly conserved aromatic residue in β7-subunit, links the specificity-determining loop (SDL) and the synergistic metal ion binding site (SyMBS) through cation-π interaction. Mutations of F185 that disrupted the SyMBS cation-F185 interaction led to deficient firm cell adhesion mediated by high affinity α4β7, and only slightly affected rolling adhesion mediated by low affinity α4β7. Disruption of SyMBS cation-F185 interaction induced partial extension of integrin ectodomain and separation of cytoplasmic tails, and impaired α4β7-mediated bidirectional signaling. In addition, loss of SyMBS cation-F185 interaction increased paxillin expression and promoted paxillin-integrin binding, leading to deficient cell spreading. Furthermore, integrin α4β7-mediated cell migration was decreased by the abolishment of SyMBS cation-F185 interaction.
This work was supported by grants from the Ministry of Science and Technology of China, the National Natural Science Foundation of China, the Chinese Academy of Sciences, and the Science and Technology Commission of Shanghai Municipality.
 Appendix:
Appendix: