α-Ketoglutarate Identified to Directly Activate NF-κB Signaling as the Second Messenger
Source:
Time: 2019-08-23
In a recent study published online in
Molecular Cell on August 22, research teams led by Dr. YANG Weiwei from Shanghai Institute of Biochemistry and Cell Biology of Chinese Academy of Sciences (CAS) and Dr. LI Guohui from Dalian Institute of Chemical Physics of CAS reported a new role of α-ketoglutarate (α-KG), a metabolic intermediate in the
glutaminolysispathway, to directly activate NF-κB signaling and promote brain tumor development.
The rapid proliferation and excessive anabolism of tumor cells, coupled with the dysregulated vasculature within the tumor, lead to the lack of nutrition in the tumor microenvironment. Tumor cells can rapidly adapt to nutritional scarcity by reprogramming their metabolic pathways, while underlying mechanism remains poorly understood.
In this study, scientists demonstrated that serine (S) 384-phosphorylated glutamate dehydrogenase 1 (GDH1) interacts with IKK complex upon glucose deficiency. GDH1 is the key enzyme in glutaminolysis pathway that converts glutamate to α-KG (Figure 1). GDH1-produced local source of α-KG directly binds to and activates IKKβ and downstream NF-κB signaling, which promotes tumor cell survival under low glucose and ultimately accelerates gliomagenesis (Figure 2).
In addition, GDH1 S384 phosphorylation correlates with
NF-κB activation, as well as the malignancy and prognosis of human glioblastoma.
These results reveal a unique role of α-KG to directly activate signal pathway as a second messenger and uncover a distinct mechanism of NF-κB activation upon nutrient stress and also establish the critical role of α-KG-activated NF-κB in brain tumor development.
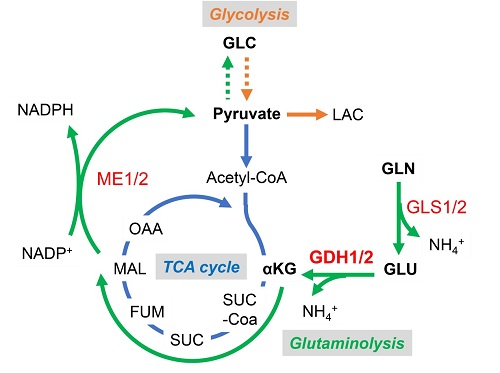
Figure 1: Schematic diagram of glutaminolysis
GLN: glutamine; GLU: glutamate; GLC: glucose.
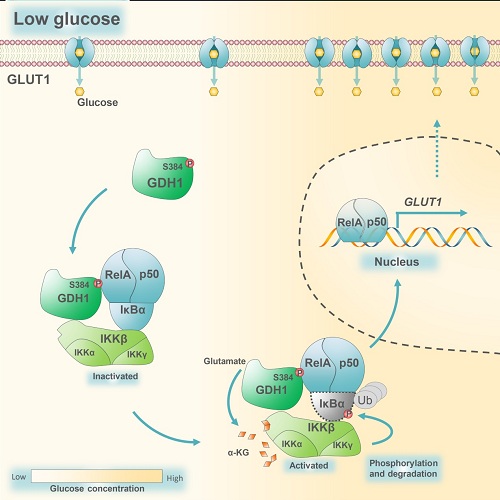
Figure 2: Schematic model of the α-KG-activated NF-κB signaling


 Appendix:
Appendix: