Scientists reveal the molecular mechanism of the membrane recruitment of the yeast TORC1-regulatory Gtr1-Gtr2 Rag GTPases by the EGO-TC complex
Source:
Time: 2019-09-26
On September 25, Science Advances published a research article entitled “Structural insights into the EGO-TC-mediated membrane tethering of the TORC1-regulatory Rag GTPases”, which is a joint research work by Prof. DING Jianping’ group at CAS Center for Excellence in Molecular Cell Science, Shanghai Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences, and Prof. Claudio De Virgilio’s group at University of Fribourg, Switzerland. This work reveals the molecular mechanism of the Ego1-Ego2-Ego3 ternary complex (EGO-TC) promoting Rag GTPase-dependent TORC1 signaling.
mTORC1 is a conserved master regulator of cell growth, proliferation, and differentiation in response to environmental conditions of energy, nutrients, and extracellular growth factors. Defects in mTORC1 signaling have been reported as causes of many severe diseases, such as cancers and diabetes. The heterodimeric human Rag and yeast Gtr GTPases serve as a key regulator in the nutrient-sensing signaling network upstream of TORC1. Analogous to mammalian Ragulator, a scaffold complex for Rag GTPases, the yeast EGO-TC interacts with Gtr1-Gtr2 to form a pentameric EGO complex (EGOC). The EGOC assembles at the surfaces of vacuoles and perivacuolar endosomes and regulates spatially distinct pools of TORC1 which phosphorylates functionally divergent effectors.
Recently, researchers from Ding’s group have revealed the molecular mechanism of the Ragulator complex promoting Rag GTPase-dependent mTORC1 signaling (Nature Communications, 2017) and also provided mechanistic insights into the structure and function of the EGO-TC in yeast TORC1 signaling (Cell Research, 2015). In this work, Dr. ZHANG Tianlong Zhang, Dr. Marie-Pierre Péli-Gulli, and their colleagues further report the crystal structure of EGOC, which shows that Ego1 wraps around Ego2, Ego3, and the C-terminal Roadblock domains of Gtr1-Gtr2 and plays a central role in the assembly of the complex.
In addition, Ego3 also interacts with both Gtr1 and Gtr2 to stabilize the complex. The functional roles of key residues involved in the assembly of the complex are validated by in vivo assays. Their structural and functional data combined demonstrate that the EGOC and the Ragulator-Rag complex are structurally conserved, and that the EGO-TC is essential and sufficient to recruit Gtr1-Gtr2 to vacuolar and endosomal membranes to ensure appropriate TORC1 signaling.
This study was supported by grants from the National Natural Science Foundation of China and the Chinese Academy of Sciences. The researchers would like to thank the staff members from BL19U1 and BL17U1 of Shanghai Synchrotron Radiation Facility (SSRF) for technical assistance in diffraction data collection.
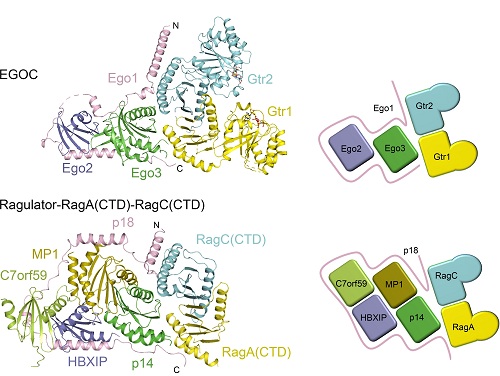
Structural comparison of the EGOC and the Ragulator-RagA(CTD)-RagC(CTD) complex.
AUTHOR CONTACT:
DING Jianping
State Key Laboratory of Molecular Biology, CAS Center for Excellence in Molecular Cell Science, Shanghai Institute of Biochemistry and Cell Biology, University of Chinese Academy of Sciences, Chinese Academy of Sciences, 320 Yue-Yang Road, Shanghai 200031, China
E-mail: jpding@sibcb.ac.cn

 Appendix:
Appendix: