Microtubules (MTs) are cytoskeletal filaments that are dynamically assembled from α/β-tubulin heterodimers. Microtubules play important roles in a variety of biological processes, such as intracellular transport, cell migration, chromosome segregation, and establishment and maintenance of cell polarity. MTs are regulated by a number of post-translational modifications on α/β-tubulin to fulfill diverse cellular functions, such as acetylation, detyrosination, glutamylation, polyamination and methylation.
Interestingly, a series of cytoskeletal proteins, such as β-actin, keratins, septins and lamin A, were found to undergo SUMOylation whereby the dynamics of actin and intermediate filaments were modulated. However, it is unclear whether MT cytoskeleton is regulated by protein SUMOylation.
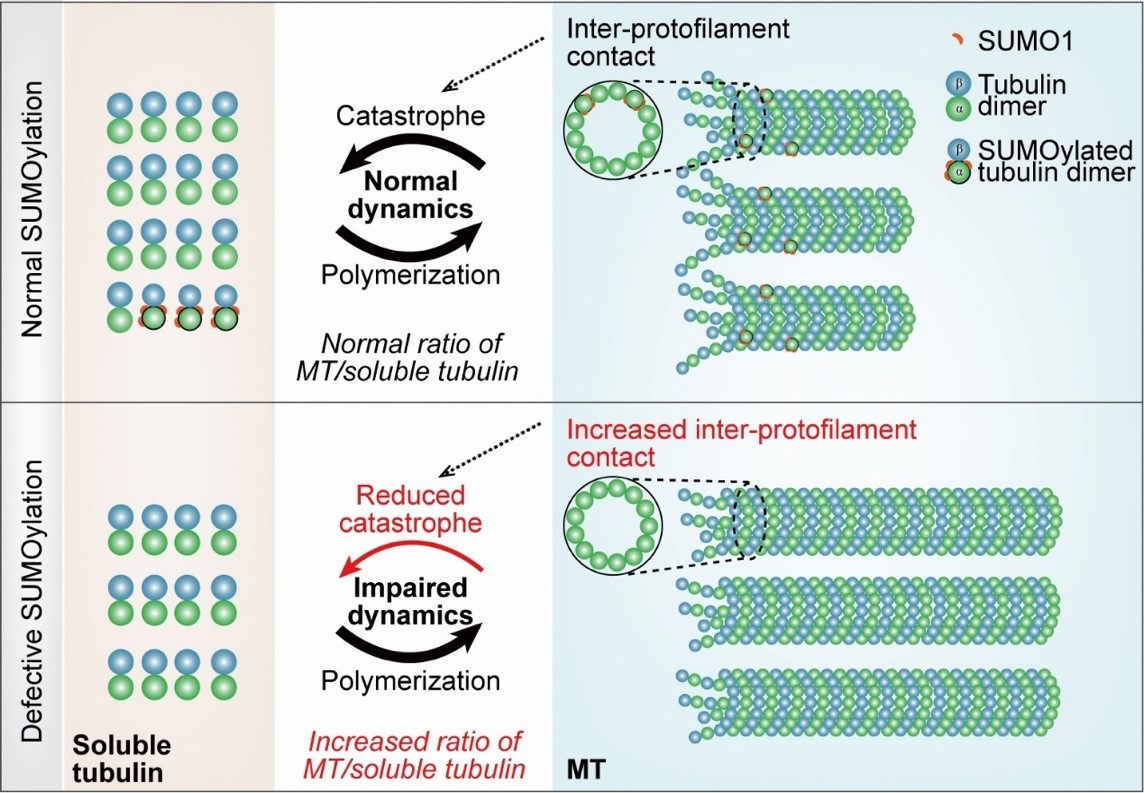
Recently, researchers from Dr. BAO Lan’s lab at the CAS Center for Excellence in Molecular Cell Science, in collaboration with Dr. LI Yong (Shanghai Jiaotong University), Dr. CHENG Jinke (Shanghai Jiaotong University) and Dr. ZHANG Xu (CAS Center for Excellence in Brain Science and Intelligence Technology), identified a novel post-translational modification on α-tubulin. The paper entitled “SUMOylation of α-tubulin is a novel modification regulating microtubule dynamics” was published in Journal of Molecular Cell Biology on May 9, 2021. The authors report that SUMOylation is a novel PTM on α-tubulin in vivo and in vitro. The SUMOylation on α-tubulin mainly occurred at K96, K166 and K304 of soluble α-tubulin and could be removed by SUMO-specific peptidase 1. Tubulin SUMOylation reduced inter-protofilament interaction, promoted MT catastrophe and impeded MT polymerization in vitro. SUMOylation of α-tubulin decreased its incorporation into MTs in cells. Thus, SUMOylation on α-tubulin represents a new player in the regulation of MT properties.
 Appendix:
Appendix: